AP 10th Class Physics 12th Lesson Magnetic Effects of Electric Current Important Questions
Class 10 Physical Science Chapter 12 Important Questions - 1 Mark
Question 1.
Mention the shape of the magnetic field lines around a current carrying straight conductor.
Answer:
In the form of concentric rings around the straight conductor.
Question 2.
State the observation made by Oersted on the basis of his experiment with current carrying conductors.
Answer:
Every current carrying conductor has a magnetic field around it.
Question 3.
State the direction of the magnetic field inside the bar magnet.
Answer:
From south to north pole.
Question 4.
Mention the special feature regarding shape of magnetic field lines.
Answer:
Magnetic field lines are closed curves.
Question 5.
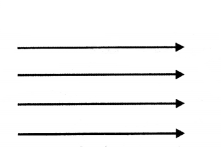
Draw a diagram to represent a uniform magnetic field in a given region.
Answer:
Question 6.
What type of core used to make an electromagnet ?
Answer:
Soft iron.
Question 7.
Identify the poles of the magnet in the given figure.
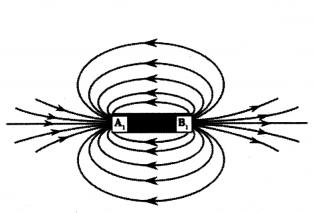
Answer:
A1 : North pole,
B1 : South pole.
Question 8.
What is the shape of magnetic field lines around a straight current carrying conductor?
Answer:
Concentric circles.
Question 9.
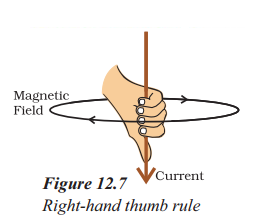
How can we find the direction of magnetic field due to a straight current carrying conductor ?
Answer:
By using right hand thumb rule.
Question 10.
What is the shape of magnetic field lines around a circular current carrying conductor?
Answer:
Straight lines at the centre of the conductor.
Question 11.
Name the rule used to find the direction of force on a current carrying conductor.
Answer:
Fleming’s left hand rule.
Question 12.
Name three colours used and names of the wires used in household wiring.
Answer:
Red, black and green (old convention) and Brown, blue and green (New convention).
Question 13.
What colour wire is used for earthing or grounding ?
Answer:
Green or yellow.
Question 14.
What is short - circuiting in an electric supply ?
Answer:
Short-circuiting in an electric circuit is a situation in which the live wire touches the neutral wire.
Question 15.
What constitutes the field of a magnet ?
Answer:
The space around a magnet with in which its magnetic effect can be experienced constitutes the magnetic field of the magnet.
Question 16.
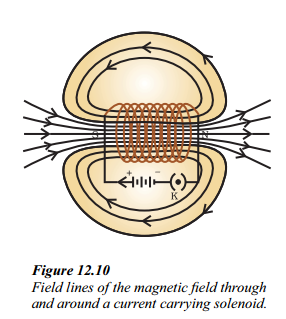
What is the shape of the magnetic field lines inside a solenoid ?
Answer:
The magnetic field lines inside a solenoid are in the form of parallel straight lines.
Question 17.
How can you magnetise a piece of magnetic material ?
Answer:
We can do so by placing it inside a current carrying solenoid.
Question 18.
How is a circuit overloaded ?
Answer:
It is overloaded by connecting too many devices to it.
Question 19.
What is short-circuiting ?
Answer:
The comming together of the line and the neutral wire is called short-circuiting.
Question 20.
How does a current carrying solenoid behave ?
Answer:
It behaves like a bar magnet.
Question 21.
What is the pattern of field lines inside a solenoid ? What do they indicate ?
Answer:
The field is in the form of parallel lines. It indicates a uniform magnetic field.
Question 22.
How is magnetic field produced in a solenoid used ?
Answer:
It is used to magnetise a soft iron piece to form an electromagnet.
Question 23.
What does the direction of thumb indicate in the right hand thumb rule ?
Answer:
The thumb indicates the direction of current in the straight conductor held by curled fingers.
Question 24.
What does the thumb indicated in Fleming’s right hand rule ?
Answer:
It indicates the direction of motion of conductor.
Question 25.
Name the device which is used to draw magnetic field lines.
Answer:
Compass needle.
Question 26.
Identify the poles of the magnet in the given figure.
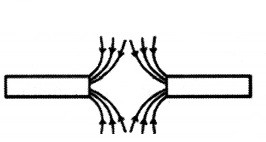
Answer:
Both the poles facing each other represent south pole in nature as the magnetic field lines outside the magnet move from North to South poles.
Question 27.
A magnet AB is broken into two pieces. What is the polarity of A, B, C and D ?

Answer:
If A is the North pole, then
C - South pole, D - North pole, B - South pole.
Question 28.
Name two parts of your body where magnetic field is produced significantly.
Answer:
Heart and Brain.
Question 29.
Suggest one way of discriminating a wire carrying current from a wire carrying no current.
Answer:
Deflection in the compass needle discriminates a wire carrying current from a wire carrying no current.
Question 30.
The diagram shows a coil of wire wound on a soft iron core forming an electromagnet. A current is passed through the coil in the directions indicated by the arrows. Mark the N and S poles produced in the iron core.
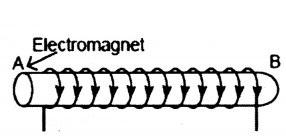
Answer:
Using clock face rule A is South pole, B is North pole.
Question 31.
What will the polarity be of one end of a solenoid if the current appears to he flowing anticlock wise in it ?
Answer:
North pole by using clock face rule.
Question 32.
What type of core should be used inside a solenoid to make an electromagnet ?
Answer:
Soft iron core.
Question 33.
What is the use of soft iron in an electromagnet ?
Answer:
It makes the magnetic field stronger.
Question 34.
Name the device used to prevent damage to the electrical appliances and the domestic circuit due to overloading.
Answer:
Electric fuse.
Question 35.
Give one difference between the wires used in the element of an electric heater and in a fuse.
Answer:
Electric wire used in electric heater has a high melting point whereas fuse wire has a low melting point.
Question 36.
A magnet is hang using a string. How will you identify the poles ?
Answer:
The north seeking pole is north pole and the south seeking pole is south pole.
Question 37.
What is the colour of wire conventionally used for (i) Live, (ii) Earth and (iii) Neutral.
Answer:
(i) Red,
(ii) Green,
(iii) Black
Question 38.
How is the fuse connected in an electric circuit ?
Answer:
Fuse is connected in series in the circuit.
Question 39.
What is the capacity of a fuse commonly used in domestic electrical fittings ?
Answer:
Fuses of capacity 5 A or 15 A are commonly used in domestic electrical fittings.
Question 40.
What is the purpose of drawing a magnetic field line with the help of a compass needle ?
Answer:
The purpose is to visually represent and understand the magnetic field around a magnet or current-carrying conductor.
Question 41.
The diagram below shows the magnetic field lines due to a current in a straight conductor.
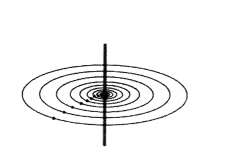
Something was done to the current because of which the magnetic field changed as shown below.
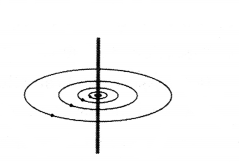
What was done to the current ?
Answer:
Its magnitude was decreased.
Question 42.
A constant current flows in a horizontal wire in the plane of the paper from east to west as shown fig. What will be the direction of magnetic field at a point North to South ?
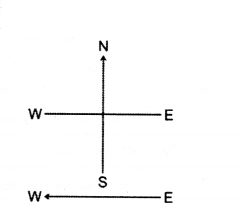
Answer:
Directly below the wire
Question 43.
A copper wire is held between the poles of a magnet. The current in the wire can be reversed. The pole of the magnet can also be changed over. In how many of the four directions shown can the force act on the wire ?
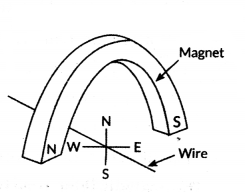
Answer:
In two directions.
Question 44.
Two statements are given below as Assertion (A) and Reason (R).
Read the statements and choose the correct option :
Assertion (A) : A current carrying straight conductor experiences a force when placed perpendicular to the direction of magnetic field.
Reason (R) : The net charge on a current carrying conductor is always zero.
a) Both (A) and (R) are true and (R) is the correct explanation of (A).
b) Both (A) and (R) are true but (R) is not the correct explanation of (A)
c) (A) is true but (R) is false.
d) (A) is false but (R) is true.
Answer:
b) Both (A) and (R) are true but (R) is not the qorrect explanation of (A)
Question 45.
Assertion (A) : The magnetic field lines around a current carrying straight wire do not intersect each other.
Reasons (R) :,The magnitude of the magnetic field produced at a given point increases as the current through the wire increases.
a) Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of the Assertion (A)
b) Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of the Assertion (A)
c) Assertion (A) is true, but Reason (R) is False.
d) Assertion (A) is false, but Reason (R) is true.
Answer:
b) Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of the Assertion (A)
Question 46.
Assertion : On freely suspending a current - carrying solenoid, it comes to rest in Geographical N-S direction.
Reason : One end of current carrying straight solenoid behaves as a North pole and , the other end as a South pole, just like a bar magnet.
a) Both A and R are true and R is the correct explanation of A.
b) Both A and R are true and R is not the correct explanation of A.
c) A is true but R is false.
d) A is false but R is true.
Answer:
a) Both A and R are true and R is the correct explanation of A.
Question 47.
Name and state the rule to determine the direction of force experienced by a current carrying straight conductor placed in a uniform magnetic field which is perpendicular to it.
Answer:
The direction of the force experienced by current carrying straight conductor placed in magnetic field which is given perpendicular to it is given by Fleming’s left hand Rule.
Question 48.
Explain why alternating current is preferred over direct current for transmission over long distance.
Answer:
A.C. can be transmitted to distant places without much loss of electric power than D.C. That is why A.C. is preferred over D.C. for transmission of current over a long distances.
Magnetic Effects of Electric Current Class 10 Important Questions - 2 Marks
Question 1.
Why do two magnetic field lines not intersect each other ?
Answer:
The two magnetic field lines do not intersect each other. If they intersect at the point of intersection, then there will be two different directions of magnetic field, which is impossible.
Question 2.
What is the direction of magnetic field lines inside and outside of a bar magnet ?
Answer:
Direction of magnetic field lines.
Inside a bar magnet : From South pole to North pole.
Outside of a bar magnet : From North pole to a South pole.
Question 3.
Sate the conclusions that can be drawn from the observation that a current carrying wire deflects a magnetic needle placed near it.
Answer:
Current carrying wire exhibits properties of magnetism, which indicate that electricity and magnetism are related phenomena.
Question 4.
How can you show that the magnetic field produced by a given electric current in the wire decreases as the distance from the wire increases ?
Answer:
The decrease in deflection of the magnetic compass needle clearly shows that the magnetic field decreases as we move away from the current-carrying conductor.
Question 5.
A current carrying straight wire held perpendicular to the plane of paper and current passes through this conductor in the vertically upward direction. What is the direction of magnetic field produced around it ?
Answer:
According to right-hand thumb rule, the direction of magnetic field produced around the given conductor is anti-clockwise.
Question 6.
If the circular coil has ‘n’ turns, the field produced is ‘n’ times as large as that produced by a single turn. Justify it.
Answer:
This is because the current in each circular turn has the same direction, and the field due to each turn then just adds up along the axis of the coil.
Question 7.
State important features of the magnetic field obtained inside the solenoid. Write one use of solenoid.
Answer:
The field lines inside the solenoid are parallel and closely spaced showing the field is highly uniform, same in strength at all the points and in the same direction.
Solenoid is used for making electromagnet.
Question 8.
What are the factors which govern the force experienced by a current carrying conductor placed in a uniform magnetic field depends ?
Answer:
The factors which govern the force experienced by conductor which is placed in a uniform magnetic field are
a) strength of the magnetic field in which conductor is placed.
b) strength of current flowing through the conductor is placed.
c) length of conductor.
Question 9.
How will the magnetic field produced at a point due to a current carrying circular . coil change if we :
a) increase the current flowing through the coil ?
b) reverse the direction of current through the coil ?
c) increase the number of turns in the coil ?
Answer:
Magnetic field (B) at the centre of the circular coil
a) increases if the current is increased as B α I
b) reverse on reversing the current.
c) increases if the number of turns in the coil increases as field is directly proportional to the number of turns.
Question 10.
How does the strength of the magnetic field produced by a current carrying solenoid increased ?
Answer:
Strength of the magnetic field can be increased by
a) increasing the current in the coil.
b) increasing the number of coils in the solenoid and
c) using a soft iron core within the solenoid.
Question 11.
State the direction of magnetic field in the case as shown
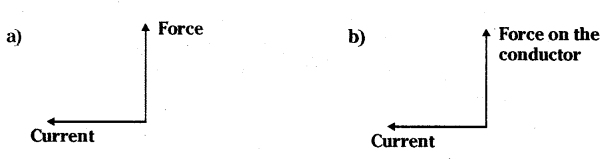
Answer:
a) In given situation, according to Fleming’s Left Hand Rule,
Force is along Y-axis.
Current is along X - axis x
Then magnetic field is along Z - axis.

b) Perpendicular to the plane of paper in the outward direction by using Fleming’s Left Hand Rule.
Question 12.
Name the material which can be used to make
i) Permanent magnet
ii) Temporary magnet
Answer:
Material which can be used to make :
i) Permanent magnet - steel, alloys (Alnico and Ni permananet magnet)
ii) Temporary magnet - soft iron.
Question 13.
State two ways by which the strength of an electromagnet can be increased
Answer:
Strength of electromagnet can be increased by
- increasing the number of turns per unit length of the solenoid and
- increasing the magnitude of the current through the solenoid.
Question 14.
What happens to the force acting on current carrying conductor placed in magnetic field when ?
a) Direction of magnetic field is reversed without changing the direction of current.
b) Direction, of the current is reversed without changing the direction of magnetic g field.
c) Direction of both the current and the magnetic field is reversed.
Answer:
Force acting on a current carrying conductor placed in a magnetic field will
a) acts in opposite direction to that of previous direction.
b) acts in opposite direction to that of previous direction.
c) remains in the same direction.
Question 15.
Wires carrying electricity should not be touched when bare-footed. Give reason.
Answer:
When we touch the live wire bare-footed, our body is directly in contact with the earth. So, current passes through the body to the earth. As our body is good conductor of electricity, we get a severe shock. Hence, we should not handle live wires bare footed.
Question 16.
Why we must not use many electrical appliances simultaneously ?
Answer:
When many high power rating appliances are switched on simultaneously, a large amount of current flows through the main circuit and current may exceed the bearing capacity of the connecting wires. This cause overloading, which may cause fire. Hence, we must not use many electrical simultaneously.
Question 17.
How is the strength of magnetic field near a straight current - conductor.
i) related to the strength of current in the conductor ?
ii) is affected by changing the direction of flow of current in the conductor ?
Answer:
i) The strength of magnetic field around a straight current conductor increases on increasing the strength of current in the conductor of vice versa.
ii) The direction of magnetic field around a straight current carrying conductor gets reversed if the direction of current through that conductor is reversed.
Question 18.
Why is earthing important for electrical appliance ?
Answer:
It safes the person operating the device from electric shock and also saves the device from excessive currents due to some fault.
Question 19.
Define a solenoid. How is it different from a coil ?
Answer:
- A coil of many circular turns of insulated copper wire wrapped closely in the shape of a cylinder is called a solenoid.
- The strength of magnetic field inside a coil vary while in case of solenoid, it is the same.
Question 20.
A compass needle is placed near a current carrying wire. State your observation for the following cases and give reasons for the same in each case.
a) Magnitude of electric current in the wire is increased.
b) The compass needle is displaced away from the wire.
Answer:
a) Deflection of compass needle increases as strength of the magnetic field is directly proportional to the amount of current.
b) Deflection of compass needle decreases as strength of magnetic field is inversely proportional to the distance from the wire.
Question 21.
Draw magnetic field lines produced around a current carrying straight conductor passing through cardboard. How will the strength of the magnetic field change, when the point where magnetic field is to be determined, is moved away from the straight wire carrying constant current ?
Justify your answer.
Answer:
The diagram is as shown
We will move a compass needle away from the straight wire. When it is moved away from the straight wire, the deflection of the needle decreases which implies the strength of the magnetic field decreases.
Question 22.
Name two safety measures commonly used in electric circuits and appliances, what precautions should be taken to avoid the over loading of domestic electric circuits?
Answer:
- Fuse - cuts off high current.
- Proper earthing.
Too many plugs should not be connected to the same socket
The load should be in accordance to the rating of the fuse in the circuit.
Question 23.
Write the two uses of magnetic compass.
Answer:
- A magnetic compass can be used to find direction.
- It can be used to test if a substance has magnetic properties.
Question 24.
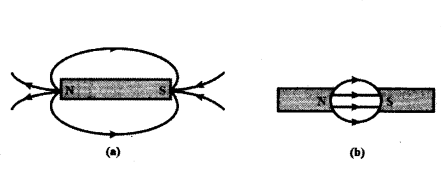
i) Identify the poles of the magnet in the figure (a) and (b) shown below.
ii) How does the strength of the magnetic field at the centre of a circular coil of a wire depend on (a) radius of the coil ? (b) number of turns in the coil ?
Answer:
i) The magnetic field lines emerge from north pole and merge at the south pole.
ii) a) More the radius weaker the field.
b) Field strength is directly proportional to the number of turns in the coil.
Question 25.
List two distinguishing features between the Solenoid and Bar magnet.
Answer:
| Solenoid |
Bar magnet |
| 1) Field disappear on stopping the current |
1) No effect of current on magnetic field. |
| 2) Strength of the field can be changed by changing the current. |
2) Strength cannot to changed. |
| 3) Direction can be reversed by charging the direction of current through it. |
3) Direction is fixed and cannot be reversed. |
Question 26.
What is the right-hand thumb rule used for ?
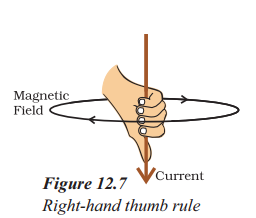
Answer:
- Imagine that you are holding a current-carrying straight conductor in your right hand such that the thumb points towards the direction of current.
- Then your fingers will wrap around the conductor in the direction of the field lines of the magnetic field, as shown in Figure. This is known as the right-hand thumb rule.
Question 27.
What are the wires called in a domestic electric circuit ?
Answer:
- Live wire, usually with red insulation cover, also known as the positive wire.
- Neutral wire, with black insulation cover, also known as the negative wire.
- Earth wire, with green insulation cover, is used as a safety measure. It is connected to a meted plate deep in the earth near the house.
Question 28.
How can the direction of the magnetic field produced by a straight wire carrying current be determined ?
Answer:
- The direction of the magnetic field can be determined by using a compass.
- Place a compass at a point near the wire and observe the direction of the needle.
- The direction of the north pole of the compass needle gives the direction of the magnetic field lines produced by the electric current through the wire at that point.
Question 29.
List two factors on which the strength of magnetic field at a point due to a current carrying straight conductor depends. State the rule that determines the direction of magnetic field produced in this case.
(OR)
State how the magnetic field produced by a straight current carrying conductor at a point depends on (a) current through the conductor (b) distance of point from conductor.
Answer:
Factors on which the magnetic field produced by a straight current-carrying conductor depends
Current in the conductor : The magnitude of the magnetic field produced is directly proportional to the current passing through the wire.
Distance from the wire : The magnitude of the magnetic field produced is inversely proportional to the distance from the wire. The Right Hand Thumb rule gives the direction of the magnetic field.
Question 30.
A compass needle is placed near a current carrying straight conductor. State your observation for the following cases and give reasons for the same in each case.
a) Magnitude of electric current is increased.
b) The compass needle is displaced away from the conductor.
Answer:
a) Observation : The deflection of the needle increases.
Reason : Magnetic field strength due to current-carrying wire increases as current in the wire increases, B α l.
b) Observation : The deflection in the compass needle decreases as its displacement
from the current-carrying wire increases.
Reason : The strength of magnetic field reduces with the increase in distance from the wire as B α l/r.
Question 31.
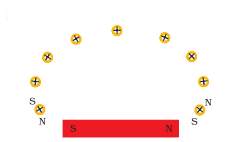
Refer to the image below and state how the magnetic field pattern indicates regions where the magnetic field is stronger outside the magnet ? What happens to the magnetic field when the current in the circuit is reversed ?
Answer:
The magnetic field strength is more than in the region where the field lines are crowded. This means the field strength is maximum near the poles and it reduces as we go away from the poles.
The direction of the magnetic field is also reversed.
Question 32.
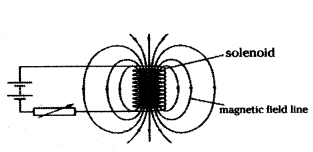
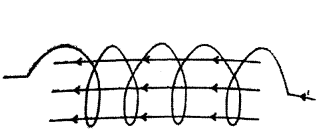
What is a solenoid ? When does a solenoid behave as a magnet ? Draw the pattern of the magnetic field produced inside it showing the direction of the magnetic field lines.
Answer:
1) Solenoid : A long coil of wire consisting of closely packed loop is called solenoid.
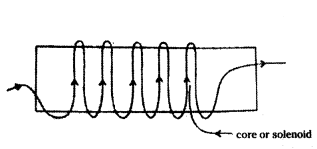
2) A solenoid acts as a magnet when current flows through it.
3) Pattern of magnetic field lines inside solenoid.
Question 33.
For the current carrying solenoid as shown, draw magnetic field lines and give reason to explain that out of the three points A, B and C, at which point the field strength is C maximum and at which point ills minimum?
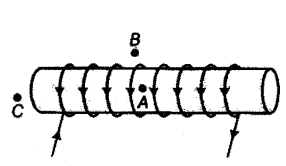
Answer:
1) Magnetic field lines through ideal solenoid is as shown below :
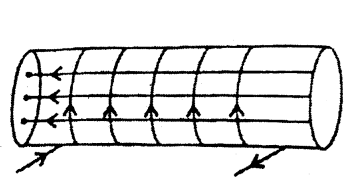
2) For an ideal solenoid, magnetic field outside the solenoid is zero and inside it is constant. So, magnetic field will be maximum at ‘B’ and is zero and minimum at ‘B’ and ‘C.
Question 34.
Draw the pattern of the magnetic field lines around a current carrying solenoid. Mark on the pattern the region where the magnetic field is uniform.
Answer:
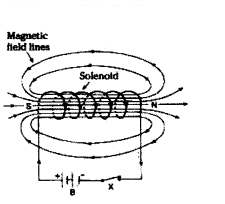
- imSolenoid is a coil of many circular turns of insulated upper wire wrapped closely in the form of cylinder.
- Uniform magnetic field inside the coil the lines of magnetic field are parallel, so magnetic field is uniform inside the coil.
Question 35.
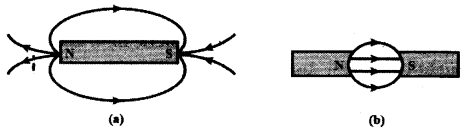
a) Name the poles P, Q, R and S of the magnets in the following figures ‘a’ and ‘b’.
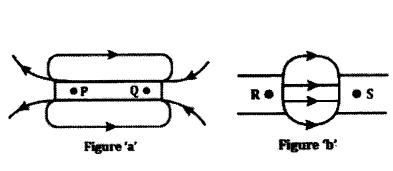
b) State the inference drawn about the direction of the magnetic field lines on the basis of these diagrams.
Answer:
a) In figure a) P - North Pole: Q - South Pole b) R - North Pole : S - South Pole
b) Magnetic field lines always starts from North Pole and end at South Pole.
Question 36.
a) In the circuit below, in case of an overload, will the fuse protect the electric oven from damage ? Justify your answer.
b) If the oven has a rating of 13 A, what should be the minimum rating of the fuse ?
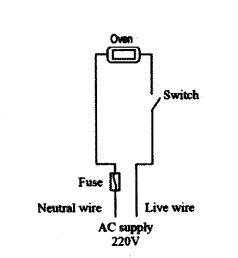
a) No.
The fuse will blow only after current has passed through the oven and damaged it.
b) a little above 13 A, (or) 14 A (or) 15 A
Question 37.
Find the direction of magnetic field due to a current carrying circular coil held :
i) vertically in North - South plane and an observer looking it from east sees the current to flow in anticlockwise direction
ii) vertically in East - West plane and an observer looking it from south sees the current to flow in anticlockwise direction,
iii) horizontally and an observer looking at it from below sees current to flow in clockwise direction.
Answer:
According to right hand rule, the direction of magnetic field is
i) west to east
ii) north to south
iii) into the paper.
Question 38.
Give reason for the following :
The burnt out fuse should be replaced by another fuse of identical rating.
Answer:
- A burnt out fuse should be replaced with identical rating because it helps in protecting the circuit from overloading and short circuiting.
- If a fuse of higher rating is used then it may not melt and cut off the supply during overloading.
- Similarly a fuse of lower rating may melt frequently even for a normal flow of current.
- This results in decreasing the efficiency of the circuit.
Question 39.
How is the direction of magnetic field at a point determined using the field lines?
Why do two magnetic field lines not cross each other ?
Answer:
- The direction of the magnetic field lines at a place can be determined by using a compass needle.
- Two magnetic field lines never intersect each other. If they would intersect, this would mean that there are two directions of the field at the specific point which is not possible.
Important Questions on Magnetic Effects of Electric Current Class 10 - 4 Marks
Question 1.
For the current carrying solenoid as shown below, draw magnetic field lines and giving reason explain that out of the three points A, B and C at which point the field strength is maximum and at which point it is minimum
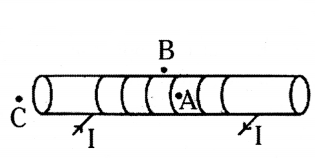
Answer:
Outside the solenoid magnetic field is minimum. At the ends of solenoid, magnetic field strength is half that of inside it. So minimum - at point B, Maximum - at point A.
Question 2.
What are the magnetic lines of force ? Give their important properties.
Answer:
Magnetic lines of force : A magnetic line of force may be defined as the curve the tangent to which at any point gives the direction of the magnetic field at that point. It may also be defined that the path along with a free north pole tends of to move.
Properties of lines of force :
- These are closed curves which start in air from the N-pole and end the S-pole and then return to the N-pole through the interior of the magnet.
- No two magnetic lines of force can intersect each other.
- They start from and end on the surface of the magnet normally.
- The lines of force have a tendency to contract lengthwise and expand side waves. This explains attraction between unlike poles and repulsion between like poles.
- The relative closeness of the lines of force gives a measure of the strength of the magnetic field which is maximum at the poles.
Question 3.
Explain with the help of the pattern of magnetic field lines the distribution of magnetic field due to a current carrying circular loop.
Answer:
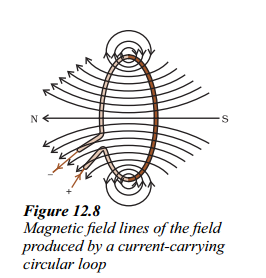
Magnetic field due to a current through a circular loop :
- Figure shows the magnetic field lines of a circular wire carrying a current.
- The lines of force near the wire are almost concentric circles.
- As we move towards the centre of loop, the concentric circles become larger and larger.
- Near the centre of the loop, the arcs of these big circles appear as parallel straight lines.
- Thus the magnetic field is almost uniform at the centre of the loop.
- By applying right hand rule, we can see that the magnetic field lines due to all sections of the wire in the same direction within the loop.
- That is why the field is stronger at the centre or near the circumference of the loop.
Question 4.
Magnetic field lines of two magnets are shown in fig (A) and (B).
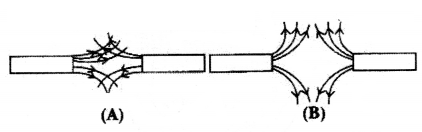
Select the figure that represents the correct pattern of field lines. Give reasons for your answer. Also name the poles of the magnets facing each other.
Answer:
Figure (B) represents the correct pattern of field lines. In figure (A), field lines cross each other which is not possible because at the point of intersection, there will be two directions of field lines.
In figure (B), field lines are emerging in nature, so poles of magnet facing each other are north poles while opposite faces will have south polarity.
Question 5.
a) Draw a schematic diagram of one of the common domestic circuits.
b) Why is it necessary to connect an earth wire to electric appliances having metallic covers ?
Answer:
a) The schematic diagram is as shown below :

b) It is necessary to connect an earth wire to electrical appliances having metallic covers to protect us from the harmful effects of electric shock, as the earth wire at the time of any leakage of current or any other defect carries the current to the earth.
Question 6.
How do iron filings align themselves near a bar magnet ?
Answer:
- Iron filings align themselves along the magnetic field lines near a bar magnet.
- The pattern created by the iron filings represents the magnetic field around the magnet.
- The iron filings arrange in a pattern due to the force exerted by the magnet.
- The region surrounding a magnet, where the force of the magnet can be detected, is called the magnetic field.
- The magnetic field lines are the lines along which the iron filings align themselves.
Question 7.
How is the strength of the magnetic field determined.
Answer:
- The closeness of the magnetic field lines.
- The degree of crowding of the magnetic field lines.
- The relative proximity of the field lines.
- The pattern in which iron filings align themselves along the magnetic field lines.
- The force exerted on a current-carrying conductor in the magnetic field.
Question 8.
How do iron filings align themselves near a current-carrying circular loop ?
Answer:
- They align themselves in a pattern of concentric circles around the loop.
- The iron filings align along the magnetic field lines produced by the current flowing through the loop.
- The direction of these concentric circles represents the direction of the magnetic field produced by the current-carrying loop.
- The pattern formed by the iron filings demonstrates the presence and shape of the magnetic field around the circular loop.
Question 9.
What is the function of the earth wire in a domestic electric circuit ?
Answer:
- The earth wire is a safety measure in domestic circuits.
- It is connected to a metal plate deep in the earth near the house.
- It provides a low-resistance conducting path for the current.
- It ensures-that any leakage of current to the metallic body of appliances keeps their potential the same as the earth.
- It prevents the user from getting a severe electric shock.
Question 10.
What is the basis for obtaining medical images using Magnetic Resonance Imaging (MRI) ?
Answer:
The basis for obtaining medical images using Magnetic Resonance Imaging (MRI) is :
- MRI uses the magnetic field produced by electric currents in the body.
- The magnetic field aligns the hydrogen atoms in the body.
- Radio waves are then directed at the aligned atoms, causing them to produce faint signals.
- These signals are detected by the MRI machine and converted into detailed images of the body.
Question 11.
What is the function of an earth wire and why is it necessary to earth metallic appliances ?
Answer:
- The function of an earth wire is to provide a path for current to flow into the ground in case of a fault or leakage in the electrical system.
- It is necessary to earth metallic appliances to ensure the safety of users.
- Earth wiring prevents the risk of electrical shocks by redirecting current away from the appliance and into the ground.
- By grounding metallic appliances, any leakage of current to a metallic body will not result in severe shocks to the user, as the current will be safely discharged into the earth.
- Earth wiring acts as a protective measure to prevent electrical accidents and damages to both the appliances and the electrical circuit.
Question 12.
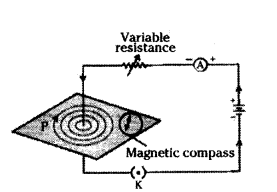
How can the deflection of a compass needle be; observed on passing an electric current through a metallic conductor ?
Answer:
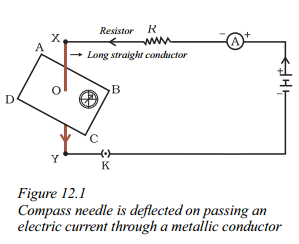
- Place a small compass near a straight copper wire carrying an electric current.
- Observe the position of the compass needle before passing the current.
- Insert the key to complete the circuit and allow the current to flow through the wire.
- Passing current through the copper wire XY causes a deflection in the compass needle.
Question 13.
How are the field lines around a bar magnet represented in Figure ?
Answer:
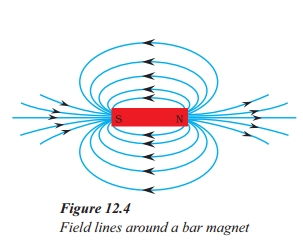
- The field lines around a bar magnet are represented by closed curves in Figure.
- The field lines emerge from the north pole of the magnet and merge at the south pole.
- The direction of the field lines is taken to be the direction in which a north pole of a compass needle moves inside the magnetic field.
- The relative strength of the magnetic field is shown by the degree of closeness of the field lines.
- No two field lines in Figure cross each other as it would result in the compass needle pointing towards two directions.
Question 14.
What happens to the deflection of the compass needle when the direction of the current in the circuit shown in Figure is reversed ?
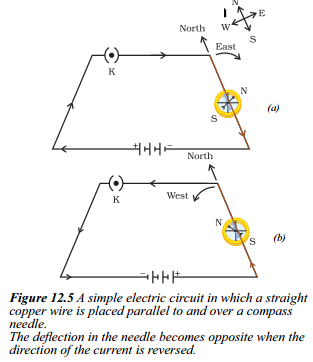
Answer:
- When the direction of the current in the circuit is reversed, the deflection in the compass needle also becomes opposite.
- In the original circuit configuration (Fig. a), where the current flows from north to south, the north pole of the compass needle moves towards the east.
- However, when the cell connections in the circuit are reversed as shown in Figure (b), resulting in the change of current direction from south to north, the needle moves in the opposite direction, towards the west.
Question 15.
What does Figure represent ?
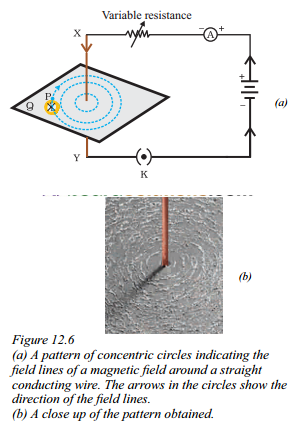
Answer:
Figure represents the pattern of the magnetic field lines around a straight conducting wire.
a) shows a pattern of concentric circles indicating the field lines of the magnetic field.
The arrows in the circles indicate the direction of the field lines.
b) a close up of the pattern obtained, providing a clearer view of the field lines.
The figure visually represents the shape and direction of the magnetic field surrounding the wire.
Question 16.
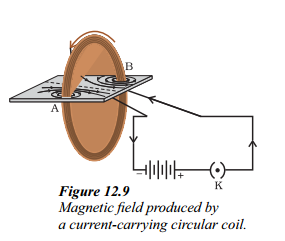
i. What is the purpose of mentioned in Figure ?
Answer:
The purpose is to observe and understand the pattern of the magnetic field produced by a current-carrying circular coil on a cardboard covered with iron filings.
ii. How is the direction of the magnetic field inside and outside the circular loop determined ?
Answer:
The direction of the magnetic field inside the circular loop is determined by applying the right-hand rule. If the current in the loop flows clockwise when viewed from above, the magnetic field inside the loop is in the anticlockwise direction. Outside the loop, the magnetic field direction is in the clockwise direction.
iii. What is the significance of the magnetic field produced by a current-carrying solenoid ?
Answer:
The magnetic field produced by a current-carrying solenoid is uniform inside the solenoid. It is used to magnetize a piece of magnetic material, like a steel rod, when placed inside the coil, creating an electromagnet. The pattern of the magnetic field lines around a solenoid is similar to that around a bar magnet.
Question 17.
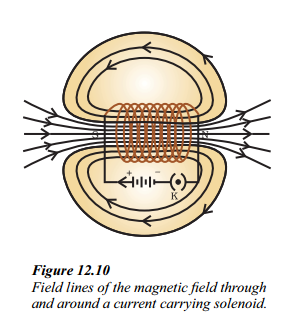
i. What does Figure depict ?
Answer:
Figure shows the field lines of the magnetic field through and around a current-carrying solenoid.
ii. What is the pattern of the magnetic field lines depicted in Figure ?
Answer:
The field lines around the current-carrying solenoid are in the form of parallel straight lines, indicating a uniform magnetic field inside the solenoid.
iii. How does the magnetic field inside a solenoid behave compared to the field around a bar magnet ?
Answer:
The pattern of the field lines inside a solenoid resembles that of a bar magnet. One end of the solenoid behaves as a magnetic north pole, while the other behaves as the south pole. The field is the same at all points inside the solenoid, indicating a uniform magnetic field.
Question 18.
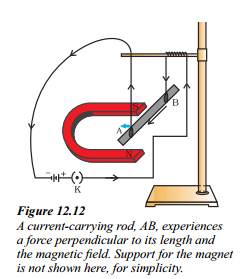
i. How does a current-carrying rod experience a force when placed in a magnetic field ?
Answer:
A current-carrying rod experiences a force that is perpendicular to both its length and the magnetic field.
ii. Does the direction of the force on the current-carrying rod depend on the direction of the current and the magnetic field ?
Answer:
Yes, the direction of the force on the conductor depends on the direction of the cur¬rent and the direction of the magnetic field.
iii. What is Fleming’s left-hand rule used for ?
Answer:
Fleming’s left-hand rule is used to determine the direction of the force acting on a current-carrying conductor in a magnetic field. According to this rule, if the first finger points in the direction of the magnetic field and the second finger in the direction of current, then the thumb points in the direction of the force.
Question 19.
Read the paragraph and answer the questions.
The magnetic effects of electric current can be observed in various phenomena. When an electric current flows through a wire, the wire behaves like a magnet. This can be demonstrated by using iron filings, which align themselves along the magnetic field lines produced by the current-carrying wire. Additionally, magnetic fields and field lines can be visualized by observing the deflection of a compass needle near a magnet, as well as the pattern formed by iron filings around a magnet. The strength of a magnetic field is determined by the closeness of the field lines and they do not cross each other. To determine the direction of a magnetic fteld, the right-hand thumb rule can be used. The magnetic field produced by a current-carrying conductor can be observed using various activities and experiments. The magnetic field produced by a current-carrying circular loop and a solenoid is similar to that of a bar magnet. Lastly, a current-carrying conductor experiences a force when placed in a magnetic field.
i. How can the magnetic effect of electric current be demonstrated using iron filings?
Answer:
Iron filings align themselves along the magnetic field lines produced by a current-carrying wire.
ii. How can the direction of a magnetic field be determined ?
Answer:
The right-hand thumb rule can be used to determine the direction of a magnetic field.
iii. What is the similarity between the magnetic field produced by a current-carrying circular loop and a solenoid ?
Answer:
The magnetic field produced by both a current-carrying circular loop and a solenoid is similar to that of a bar magnet.
iv. What happens to a current-carrying conductor placed in a magnetic field ?
Answer:
A current-carrying conductor experiences a force when placed in a magnetic field.
Question 20.
Read the paragraph and answer the questions.
Magnetic fields exist in the region surrounding a magnet where the force of the magnet can be detected. Magnetic field lines represent the magnetic Held and show the direction a north pole would move inside the field. The field lines are closer together where the field is stronger. When current flows through a conductor, a circular magnetic field is produced around the conductor. The direction of this field is determined by the right-hand rule - if you point your thumb in the direction of the current, your fingers curl in the direction of the magnetic field. The magnetic field produced by a current-carrying solenoid is similar to that of a bar magnet, with parallel field lines inside the solenoid.
i. How do magnetic field lines indicate the direction of the magnetic field ?
Answer:
Magnetic field lines indicate the direction of the magnetic field. The lines point in the direction a north pole would move inside the field.
ii. How does current flowing in a conductor produce a magnetic field? Describe the right-hand rule.
Answer:
When current flows through a conductor, it produces a circular magnetic field around it. Using the right-hand rule, if you point your thumb in the direction of current flow, your fingers will curl in the direction of the magnetic field.
iii. Compare the magnetic fields produced by a current-carrying solenoid and a bar magnet.
Answer:
The magnetic field inside a current-carrying solenoid is similar to that of a bar magnet the field lines are parallel inside the solenoid, with one end acting like the north pole and the other end acting like the south pole.
iv. If the current through a conductor is increased, how will it affect the magnetic field around it ? Explain.
Answer:
Increasing the current through a conductor will increase the strength of the magnetic field around it. The field lines will become more concentrated as the field strength increases.
Question 21.
Draw the pattern of magnetic field lines produced around a current carrying straight - conductor passing perpendicular to a horizontal cardboard. State right-hand thumb rule to mark the direction of the field lines. How will the strength of the magnetic field change when the point where magnetic field is to be determined is moved away from the straight conductor ? Give reason to justify your answer.
Answer:
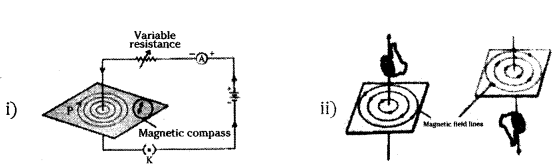
Direction of magnetic field easily found by applying the Right hand Thumb Rule. According to this rule. If we hold the current carrying conductor in right hand such the thumb is stretched along the direction of current, then fingers will wrap around . , the wire in the direction of magnetic field.
iii) The strength of magnetic field decreases as the point where magnetic field is to be determined is moved away from the straight conductor.
Question 22.
Draw the pattern of the field lines of the magnetic field around a current carrying straight conductor passing through and held perpendicular to a horizontal cardboard. State right-hand thumb rule and explain how this rule is useful to determine the direction of the magnetic field in the above case, if the direction of current in the conductor is vertically downwards.
Answer:
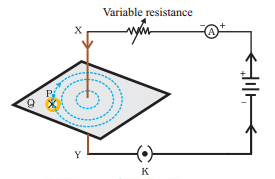
Maxwell Right hand Thumb Rule is used.
It states that suppose you are holding a current carrying conductor in your right hand in such a way that your thumb points in the direction of the current.
Then the way on direction in which your fingers will encircle will give the direction of magnetic field lines.
As in this case, the current flows downwards, so the magnetic field lines go clockwise.
finger encircle clockwise
Question 23.
Why is an alternating current (A.C.) considered to be advantageous over direct current (D.C.) for the long distance transmission of electric power ?
Answer:
- Power wasted only depends on resistance and current in the circuit and does not depend on whether its AC or DC.
- If P is the power transmitted, and R is the resistance of the line: P = IV
- Power loss = I2R = V2R
- It is better to have lower current and higher voltage to reduce losses.
- AC is much easier (therefore cheaper) to step up and down in voltage (using a transformer) than DC.
- It can be done more efficiently than the much more expensive and complex DC step-up/step-down systems.
- That is why AC is used for power transmission.
Question 24.
Study the diagram given and answer the questions that follow :
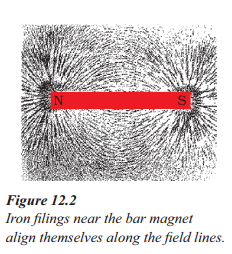
a) Why do the iron filings arrange in such a pattern ?
b) What does this pattern demonstrate ?
c) Why do the iron filings hear the bar magnet seem to align in the shape of closed curves ?
Answer:
a) This is due to the reason that each piece of iron filling becomes a small magnet and experiences a force in a certain direction in the magnetic field due to the magnet.
b) The pattern is demonstrated that the magnetic field is the strong at poles of the magnet and the magnetic field becomes weaker if we goes far from the magnet. Magnetic filed lines emerge from the north pole of the magnet and ends at the south pole of the magnet.
c) They experience magnetic force when kept near a bar magnet. The force thus exerted makes iron filings to arrange in a pattern. The region surrounding a magnet, in which the force of the magnet can be detected, is said to have a magnetic field.
Question 25.
Diagram shows the lengthwise section of a current carrying solenoid (×) indicates current entering into the page, (.) indicates current emerging out of the page. Decide which end of the solenoid A or B, will behave as north pole. Give reason for your answer.
Also draw field lines inside the solenoid.
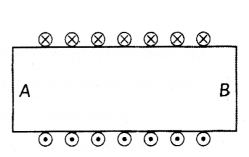
Answer:
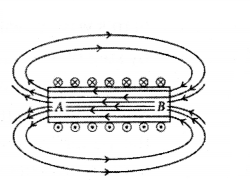
- Using right hand thumb rube we can draw the magnetic field lines around the conductor as shown.
- From figure, end A of solenoid act as north pole and end B will act as south pole. Inside the solenoid field lines are in the form of parallel straight lines.
Question 26.
The diagram below is a schematic diagram of a household circuit. The house shown in the diagram has 5 usable spaces where electrical connections are made. For this house, the mains have a voltage of 220 V and the net current coming from the mains is 22 A.
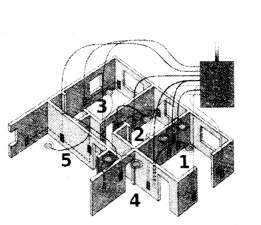
a) What is the made of connection to all the spaces in the house from the mains ?
b) The spaces 5 and 4 have the same resistance and spaces 3 and 2 have respective resistances of 20 12 and 30 12. Space 1 has a resistance double that of space 5. What is the net resistance for space 5 ?
c) What is the current in space 3 ?
d) What should be placed between the main connection and the rest of the house’s electrical appliances to save them from accidental high electric current ?
Answer:
a) All spaces arc connected in parallel.
b) Let resistance of space 5 and 4 be R ohms respectively.
Resistance of space 1 = 2 R ohms ; Resistance of space 2 = 30 ohms
Resistance of space 3 = 20 ohms
Current = 22 A : V = 2 20V: Total resistance - V/I
1R1 + 1R2 + 1R3 + 1R4 + 1R5 = 1Req ? 12R + 130 + 120 + 1R + 1R = 1Req ? R = 150 ?
c) Current in space 3.I3 = 220/20 = 11A
d) A fuse wire should be placed between the main connection and rest of the house’s electrical appliances to save them from accidental high electric current.
Question 27.
How are the magnetic field lines of a bar magnet drawn using a small compass needle ? Draw one magnetic field line each on both sides of the magnet.
Answer:

Magnetic field lines can be mapped out using a small compass.
- We will mark the position of two ends of the needle of the compass.
- Now we will move the needle to a new position such that its south pole occupies the position previously occupied by its north pole.
- In this way, we will proceed step by step till we reach the south pole of the magnet as shown in Fig.
- Now we will join the points marked on the paper by a smooth curve. This curve represents a field line.
- We will repeat the above procedure, and get the magnetic field lines around the magnet.
Question 28.
State whether an alpha particle will experience any force in a magnetic field if (alpha particles are positively charged particles)
i) it is placed in the filed at rest.
ii) it moves in the magnetic field parallel to field lines.
iii) it moves in the magnetic field perpendicular to field lines Justify your answer in each case.
Answer:
i) No, alpha particle will not experience any force if it is at rest, because only moving charge particle can experience force when placed in a magnetic field.
ii) No, alpha particle will not experience any force if it moves in the magnetic field parallel to field lines because charge particle experiences force only when it moves at an angle other than 0° with magnetic field.
iii) Alpha particle will experience a force in the direction perpendicular to the direction of magnetic field and direction of motion of alpha particle.
Extra Questions on Magnetic Effects of Electric Current Class 10 - 8 Marks
Question 1.
What is the direction and pattern of the magnetic field produced by a current¬carrying wire ?
Answer:
- The magnetic field forms circular loops around the wire.
- The direction of the magnetic field is determined by the right-hand thumb rule, where if you point your right thumb in the direction of the current, your fingers will wrap around the wire in the direction of the magnetic field lines.
- The magnetic field lines become larger and larger as you move away from the wire, forming concentric circles around it.
- The strength of the magnetic field is determined by the closeness of the field lines, with closer lines indicating a stronger magnetic field.
- The field lines do not cross each other, as that would result in the compass needle pointing towards two different directions, which is not possible.
- The north pole of a compass needle is deflected towards the east when the current flows from north to south, and towards the west when the current flows from south - to north.
Question 2.
How can the direction of the magnetic field inside and outside a current-carrying circular loop be determined Using the right-hand rule ?
Answer:
- The right-hand thumb rule can be used to determine the direction of the magnetic field.
- Place the circular loop on a table and visualize a current passing through it in a clockwise direction.
- Curl the fingers of your right hand in the direction of the Current.
- The magnetic field inside the loop will be in the direction pointed by your thumb.
- Outside the loop, the magnetic field will be in the opposite direction to that inside the loop.
- This rule helps to determine the direction of the magnetic field around a circular loop carrying current.
Question 3.
How is the direction of the magnetic field determined using a compass ?
Answer:
- Place the compass near the north pole of the magnet.
- Observe the behaviour of the compass needle.
- The south pole of the compass needle points towards the north pole of the magnet, while the north pole of the compass is directed away from the north pole of the magnet.
- Mark the position of the two ends of the compass needle.
- Move the compass needle to a new position such that its south pole occupies the position previously occupied by its north pole.
- Repeat this process step by step until you reach the south pole of the magnet.
- Join the points marked on the paper by a smooth curve. This curve represents a field line.
- These lines Represent the magnetic field around the magnet, and they are known as magnetic field lines.
- The direction of the magnetic field is taken to be the direction in which a north pole of the compass needle moves inside it. Therefore, the field lines emerge from the north pole of the magnet and merge at the south pole. Inside the magnet, the direction of field lines is from its south pole to its north pole.
- By observing the deflection of the compass needle along the field line, the deflec-tion increases as the needle is moved towards the poles.
- It is important to note that no two field lines are found to cross each other.
Question 4.
How can magnetic field lines be drawn for a bar magnet ?
Answer:
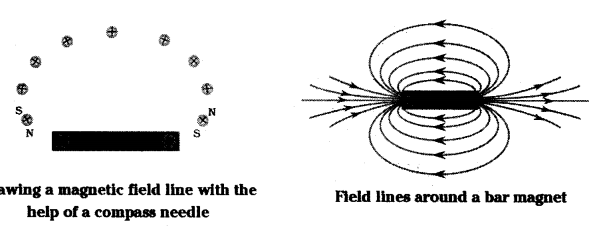
- Magnetic field lines around a bar magnet can be drawn using iron filings.
- Place the bar magnet on a sheet of white paper fixed on a drawing board.
- The iron filings will align themselves along the magnetic field lines.
- Mark the boundary of the magnet.
- Place a compass near the north pole of the magnet.
- Observe the behaviour of the compass needle.
- Mark the position of the compass needle’s ends.
- Move the compass needle to a new position such that its south pole occupies the position previously occupied by its north pole.
- Repeat this process to create multiple field lines.
- Join the marked points on the paper by a smooth curve to represent a field line.
- Draw as many field lines as possible to get a pattern representing the magnetic field around the magnet.
Question 5.
How is electric power supplied to our homes ?
Answer:
- Electric power is supplied to our homes through a main supply, either supported through overhead electric poles or by underground cables.
- The main supply consists of two wires: the live wire (or positive) with red insula¬tion cover and the neutral wire (or negative) with black insulation cover.
- The potential difference between the live wire and the neutral wire is typically 220 V.
- At the meter-board in the house, the wires pass into an electricity meter through a main fuse.
- The wires are then connected to the line wires in the house, which supply electricity to separate circuits within the house. Often, two separate circuits are used, one with a higher current rating for appliances with higher power ratings and another with a lower current rating for bulbs, fans, etc.
- The earth wire, with green insulation, is usually connected to a metal plate deep in the earth near the house. It is used as a safety measure, especially for appliances with a metallic body, to prevent severe electric shocks.
Question 6.
A current carrying conductor is placed in a magnetic field. Now answer the following. v
i) List the factors on which the magnitude of force experience by conductor depends.
ii) When is the magnitude of this force maximum ?
iii) State the rule which helps in finding the direction of motion of conductor.
iv) If initially this force was acting from right to left, how will the direction of force change if:
a) direction of magnetic field is reversed ?
b) direction of Current is reversed ?
Answer:
i) When a current carrying wire is placed in a magnetic field, it experiences a magnetic force that depends on
a) current flowing in the conductor
b) strength of magnetic field
c) length of the conductor
d) angle between the element of length and the magnetic field.
ii) Force experienced by a current carrying conductor placed in a magnetic field is largest when the direction of current is perpendicular to the direction of magnetic field.
iii) The rule used in finding the direction of motion of the conductor placed in a magnetic field is Flemings left hand rule.
Flemings left hand rule is as follows : Stretch out the thumb, the forefinger, and the second (middle) finger of the left hand so that these are at right angles to each other. If the forefinger gives the direction of the magnetic field (N to S), the second (middle) finger the direction of current then the thumb gives the direction of the force acting on the conductor.
iv) a) Direction of force will be reversed when direction of magnetic field is reversed, i.e., now force on conductor will act from left to right.
b) Direction of force will be reversed, if the direction of current is reversed, i.e., the force on the conductor will act from left to right.
Question 7.
PQ is a current carrying conductor In the plane of the paper as shown in the figure here.
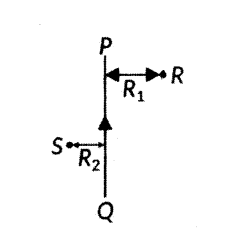
i) Find the directions of the magnetic fields produced by it at points R and S.
ii) Given R1 > R2, where will the strength of the magnetic field be larger? Give reasons.
iii) If the polarity of the battery connected to the wire reversed, how would the direction of the magnetic field be changed?
iv) Explain the rule that is used to find the direction of the magnetic field for a straight current conductor.
Answer:
i) According to right hand thumb rule, the magnetic field lines produced is: Into the plane of the paper in at R. Out of it at S.
ii) Magnetic field strength for a straight current carrying conductor is inversely proportional to the distance from the wire. So, as distance from the conductor decreases, magnetic field strength increases. Since r2 < r1 Magnetic field strength at S will be greater than magnetic field strength at R.
iii) If the polarity of the battery connected to the wire is reversed, then the direction of current through the conductor is also reversed i.e., current will now flow from top to bottom and the direction of magnetic field lines will also be reversed.
Now, the magnetic field lines will be: Out of the plane of the paper at R. Into the plane of the paper in at S.
iv) Right Hand Thumb rule is used to find the direction of the magnetic field for a straight current carrying conductor. It states that, if we point the thumb of our right hand in the direction of the current, then the direction in which our fingers curl gives the direction of the magnetic field.

Question 8.
Read the case given below carefully:
A student was asked to perform an experiment to study the force on a current carrying conductor in a magnetic Held. He took a small aluminium rod AB, a strong horse shoe magnet, some connecting wires, a battery and a switch and connected them as shown. He observed that on passing current, the rod gets displaced. On reversing the direction of current, the direction of displacement also gets reversed. On the basis of your understanding of this phenomenon.
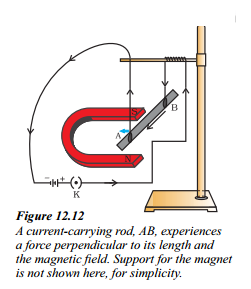
Answer the following questions:
a) Why does the rod get displaced on passing current through it ?
b) State the rule that determines the direction of the force on the conductor AjB.
c) i) In the above experimented set-up, when current is passed through the rod, it gets displaced towards the left. What will happen to the displacement if the polarity of the magnet and the direction of current both are reversed ?
ii) Name any two devices that use current carrying conductors and magnetic field.
d) Draw the pattern of magnetic field lines produced around a current carrying straight conductor held vertically on a horizontal cardboard. Indicate the direction of the field lines as well as the direction of current flowing through the conductor.
Answer:
a) The rod get displaced on passing current through it because a force is exerted on the rod when it placed in a magnetic field.
b) Fleming’s left hand rule determines the direction of the force on the conductor AB. According to this rule, stretch the thumb, forefinger and middle finger of your left hand such that they are mutually perpendicular. If the first finger points in the direction of magnetic field and the second finger in the direction of current, then the thumb will point in the direction of motion or the force acting on the conductor.
c) i) If the polarity of the magnet and the direction of current both are reversed, the rod gets displaced towards the right.
ii) Two devices that use current carrying conductors and magnetic field are electric motor and electric generator.
d) Pattern of magnetic field lines produced around a current carrying straight conductor held vertically.
Also indicated the direction of field lines as well as the direction of current flowing through the conductor.